Research
The origination and diversification of novel genital structures
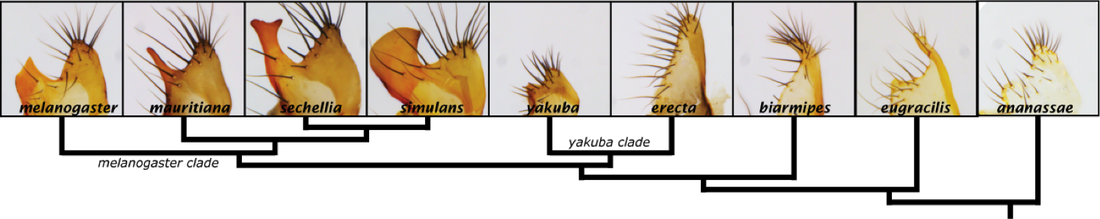 The posterior lobe is a novel hook-shaped outgrowth unique to the lateral plate of Drosophila melanogaster and its closest relatives
The posterior lobe is a novel hook-shaped outgrowth unique to the lateral plate of Drosophila melanogaster and its closest relatives
One of the biggest mysteries that the evo-devo field hopes to solve is the origins of "novelties", complex morphological structures whose beginnings are not yet clear. How does the genetic program of a complex anatomical formation evolve? We know that much of what goes into erecting a morphological structure is the patterning of gene expression through vast gene regulatory networks that control when and where genes that confer unique cellular behaviors are activated. In a sense, the question of how novelties evolve can be re-phrased to how does a new morphological structure's gene regulatory network arise? To address these questions, we have focused on recently evolved novelties because it allows us to perform careful comparisons between species that don't have these structures with their close relatives that do. We have accordingly focused on the most rapidly evolving morphological structures one can find in nature: animal genitalia. The genetic powerhouse organism, Drosophila melanogaster, while not normally considered to have the most unique and elaborate morphology, boasts a gentalia that is quite different from its closest relatives. The posterior lobe is a novel hook-shaped cuticular structure that protrudes from the genital arch and is absolutely necessary for genital coupling during mating. Our first paper on this structure's evolution described how many of the genes involved in posterior lobe development were co-opted from another structure, the posterior spiracle, an embryonic extension of the tracheal system. Thus, a large portion of a network used in a seemingly unrelated structure, has been re-deployed to generate an elaborate novelty. Our next steps are focused on finding the factors that have specifically been altered to initiate this spectacular event. In support of this goal, we have released a database of the expression patterns for the top transcription factors expressed in the genitalia. Browse our data at: https://flyterminalia.pitt.edu/!
The diversification and morphogenesis of novelties
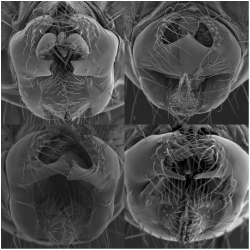 Scanning EM images of melanogaster clade genital morphologies
Scanning EM images of melanogaster clade genital morphologies
While the we now have many examples of how genetic changes alter morphological phenotypes, one area that is currently understudied is the molecular basis for the evolution of shape. What types of genes contribute to differences in anatomical shape, and how do they affect the process of tissue morphogenesis? While studying the origins of the posterior lobe, we have become interested in finding the genetic changes that underlie variation in its morphology. This has led to some surprising complexities of the system (read about it here!). In parallel, there is good evidence that changes in the posterior lobe's morphology were subject to positive selection, and we are interested in connecting the changes we have found to the signatures left by natural/sexual selection on the genome sequences of populations in which they are embedded.
The evolution of coloration traits in the Drosophila abdomen
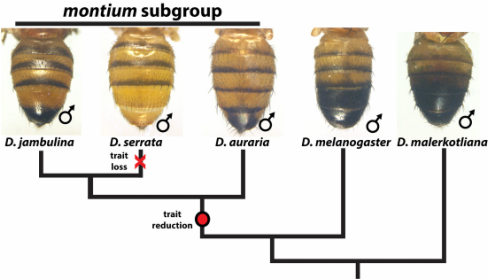 Rapidly evolving pigmentation patterns in males of the melanogaster species group
Rapidly evolving pigmentation patterns in males of the melanogaster species group
The highly variable pigmentation patterns that adorn the abdomen of Drosophila species represents one of the most rapidly evolving characters in this group of animals. The genetic basis of these traits have been studied for over a hundred years now, as mutations affecting body coloration were among the first genetic markers in the fly room of T.H. Morgan. We are currently dissecting various intra- and interspecific traits down to their causative loci at the resolution of individual nucleotide changes in the DNA, These include expansions, gains and losses of pigmentation and modification of body plan-patterning Hox genes (see our recent paper in Current Biology!). One exciting direction that we are currently cultivating is the evolution of the landscape of transcription factors of the Drosophila abdomen, and connecting these factors to their DNA targets to grasp fundamental principles of how the encoding of regulatory logic that determines gene expression evolves.
The evolutionary origins of gene regulatory sequences
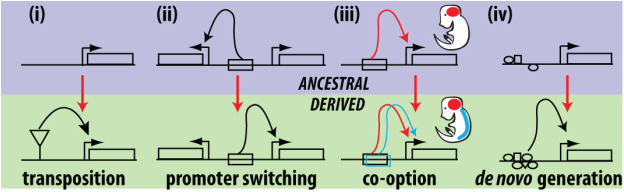 Potential mechanisms for the origins of new enhancers that drive novel expression patterns
Potential mechanisms for the origins of new enhancers that drive novel expression patterns
It has now been quite extensively demonstrated that changes in gene expression, via the alteration of gene regulatory sequences, is a common mechanism to generate morphological diversity. What is much less well understood is how new gene expression patterns evolve? This is an important question because many examples of morphological novelty appear to involve the evolution of new expression patterns. This problem can be boiled down to the question: where do gene regulatory sequences come from? Our lab has been looking for examples of recently evolved expression patterns in order to elucidate the molecular paths by which the transcriptional control sequences that generate these patterns arise. At every corner, it seems that different mechanisms are at work, and our investigations promise to reveal many new insights to this problem. This research sets the stage for evaluating the range of possibilities when trying to understand evolutionary events that occurred in the distant past.
